Application of nanoparticles to control plant pathogens is a rapidly emerging field in crop disease management, and economical and environmentally friendly nanoparticle synthesis methods are being extensively studied. . In this project, we investigated the potential of nano silver (AgNPs) synthesized by an aqueous extract of Artemisia absinthium against a number of species of Phytophthora spp., Causing many potentially important plant diseases. economy. In in vitro dose response tests conducted on microtiter plates, 10 µg ml −1 AgNP inhibited mycelium growth of P. parasitica, P. infestans, P. palmivora, P. cinnamomi , P. Tropicalis, P. capsici and P. katsurae. Detailed in vitro dose response analyzes performed with P. parasitica and P. capsici showed that AgNP synthesized with a highly effective A. absinthium extract (IC50: 2.1 to 8.3 µg ml -1) and (100%) effective in inhibiting mycelium growth, germination of animal spores, germ duct elongation and cytogenesis. Interestingly, the nano silver treatment increased the rate of coagulation in mammals. Consistent with in vitro results, in vitro planting experiments conducted in greenhouses showed that AgNP treatments prevented Phytophthora infection and improved plant survival. Furthermore, AgNP in plant experiments did not produce any adverse effects on plant growth. These studies provide a simple and economical way to control Phytophthora with AgNP without affecting normal plant physiology.

Nội dung bài viết
INTRODUCE
Nano silver (AgNPs) exhibit strong antibacterial, antifungal and anti-tumor activities (Kharissova et al. 2013). Due to their excellent antimicrobial activities and desirable physicochemical properties, AgNPs are currently being extensively studied for application in various disciplines including medicine, diagnostics, cosmetics and food processing (Thorley and Tetley 2013, Project on Nanotechnology Emerging 2013). In fact, they have been used in wound dressings, food packaging, and in consumer products such as textiles and footwear to fight odor-causing microorganisms (Schluesener and Schluesener 2013; Velmurugan et al. 2014 Project on Nanotechnology Emerging 2013). AgNP is mainly composed of zero valence silver clusters (Ag 0), typically 5 to 100 nm in diameter. Depending on their synthetic chemistry, AgNP preparations can include nanospheres, nanotubes, triangular crystals, or a combination of these shapes. The three-dimensional nanostructures of AgNPs are stabilized by various capping agents, mainly biofilm-forming agents such as cellulose, pectin, guar gum and polyethylene glycol (George et al. 2014; Lavorgna et al. 2014. ; Mandal et al. 2012; Raghavendra et al. 2013). Several mechanisms have been proposed for the antibacterial properties of AgNPs. They exhibit a high affinity for sulfur and phosphorus. Their interactions with sulfur-containing amino acids inside or outside the cell affect cell viability (Prathna et al. 2011). Another possible mechanism involves the release of silver ions from AgNP and their subsequent interaction with phosphorus in DNA, thereby inactivating DNA replication. Released silver ions can also react with sulfur-containing proteins, leading to inhibition of enzyme and protein functions (Gupta 1998). In addition, silver ions have been reported to inhibit respiratory chain proteins and interfere with membrane permeability (Holt and Bard 2005; Shrivastava et al. 2007). Since AgNPs display multiple inhibitory activity regimes against microorganisms (Clement and Jarrett 1994), the risk of pathogens developing against AgNPs is minimized. Due to reduced resistance, AgNPs can be used to more effectively control fungicide-resistant plant pathogens.
Nano silver synthesized using plant extracts have been shown to inhibit plant pathogens and bacteria (Kaur et al. 2012; Kim et al. 2012). ; Panacek et al. 2009; Pimprikar et al. 2009). However, to our knowledge, no studies have reported AgNP’s ability against oocytes, which are biologically different from real fungi. The genus oomycete Phytophthora includes more than one hundred species, which infect a wide variety of regulated agricultural food, feed and ornamental plants and forest plants in natural ecosystems (Kroon et al. 2012). Worldwide crop damage from Phytophthora disease is estimated to be billions of dollars (Wawra et al. 2012). Prominent examples include $ 6.7 billion in damage to potatoes caused by mildew (Haverkort et al. 2008) and $ 1 to $ 2 billion in soybeans due to Phytophthora root rot (Tyler 2007). Some of the most destructive and well known Phytophthora species are P. infestan s (potato and tomato blight), P. parasitica (blight, root and stem rot in ornamental and annual plants. including citrus), P. capsici (blight of many vegetables), P. sojae (soybean root and stem rot), and P. ramorum (sudden oak death) (Cacciola and Lio 2008; Cline et al 2008; Erwin and Ribeiro 1996; Gevens et al. 2007; Grunwald et al. 2012; Kroon et al. 2012). Currently, many different synthetic chemicals are used to control these pathogens. However, Phytophthora spp. is known to develop very rapid chemical resistance (Childers et al. 2015; Dobrowolski et al. 2008; Gisi and Cohen 1996; Hu et al 2012; Hu et al. 2005; Hu et al 2008, 2010; Hwang and Benson 2005; Meng et al. 2011; Perez-Sierra et al. 2011; Randall et al. 2014; Timmer et al. 1998). Fungicidal resistance is one of the major problems in disease management caused by Phytophthora spp. Addressing fungicide resistance will require the discovery of alternative products with new modes of action. AgNPs, due to the lower risk of developing resistance, may play an important role in the management of the fungicide resistance of Phytophthora spp.
AgNPs can be synthesized using a variety of physical methods such as plasma catalysis and laser resection (Amendola et al. 2007) and chemical methods requiring reducing agents such as sodium borohydrate (Borase et al. 2014). ; Zhu et al 2000). The physical method involves high temperatures and lasers, is expensive and requires specialized equipment and highly skilled personnel. Chemical synthesis methods are often associated with high temperature and pressure, and the creation of potentially dangerous byproducts poses environmental concerns (Borase et al. 2014; Zhu et al. 2000 ). Green synthesis, using biological materials as a reducing agent and capping agent, offers a relatively safer and environmentally friendly approach to nanoparticle synthesis (Borase et al. 2014). Multiple biological sources including plant and bacterial tissue extracts have been used for the synthesis of AgNPs (evaluated in Borase et al. 2014; Kharissova et al. 2013). Although the exact mechanism of how biological materials mediate AgNP synthesis is not well understood, different metabolites and enzymes are proposed to provide reduction ability to reduce Ag + to Ag 0. In addition Biological reductants and extracts have also been proposed to provide capping agents to stabilize AgNPs (reviewed in Borase et al. 2014; Kharissova et al. 2013). Artemisia absinthium L. is found naturally in the foothills of Himalayan mountains in the Indian subcontinent. Previously, our team as well as others have shown that this plant has strong antioxidant activity, providing an excellent source of Ag + to AgNP reduction (Ali and Abbasi 2014a, b; Ali et al. 2013; Lee et al. 2013; Singh et al. 2012). Using the aqueous extract of A. absinthium, we have successfully synthesized and characterized AgNP (Ali et al., Unpublished data). In this report, we investigated the potential of these AgNPs in the inhibition of Phytophthora spp. To the best of our knowledge this is the first report showing effective control of Phytophthora spp. using silver nanoparticles. The main objectives of this study were (i) to evaluate the efficacy and potency of Artemisia -mediated AgNP against different Phytophthora spp. in vitro and in planta, and (ii) to determine the effect of AgNP on different stages of development and reproduction of Phytophthoraspp. The results of our investigation will expand our portfolio of products that can be used alone or alternately with other chemicals to control Phytophthora disease.
MATERIALS AND METHODS
1. Synthesis of nano silver
Greenhouse-grown A. absinthium plants were dried at room temperature and used to synthesize AgNPs. Plant extracts are prepared by boiling 1 g of the dried powdered leaves of A. absinthium in 10 ml of deionized water for 5 minutes. The aqueous extracts are cooled to room temperature (25 ° C), filtered through a 0.45 μm filter (Millex) and stored at 4 ° C until use. For the synthesis of silver nanoparticles, AgNO3 (2 mM) and the water plant extracts prepared as described above were mixed in equal volumes, and the reactions were carried out at room temperature for 24 hours. AgNO 3 is unreacted and the plant extracts are removed by pelleting and washing AgNP as follows. The reaction mixture was centrifuged at 14,000 × g for 10 minutes at room temperature. The supernatant was removed and the AgNP pellets were re-immersed in deionized water, then centrifuged at 14,000 × g for 10 minutes. This process was repeated five times. The obtained AgNP is re-immersed in deionized water and used in antibacterial tests.
Nano silver synthesis was monitored by recording UV-vis spectra (λ 250 to 700 nm) using either the NanoDrop 2000C spectrometer (Thermo Fischer Scientific) or the Synergy H1 Hybrid multi-mode microfiber reader (BioTek).
Tests and analysis of Phytophthora inhibition data in vitro.
The antibacterial efficacy and antimicrobial efficacy of nano silver were tested against different Phytophthora spp. in vitro. Sources of Phytophthora spp. The target used in this report is provided in Table 1. However, most of the suppression trials focused on P. parasitica and P. capsici. Initially, the effects of 10-fold dilutions of AgNPs (100, 10 and 1 µg ml -1) were tested against different Phytophthora spp. In vitro tests were performed in a previously described high flux microtiter plate test (Ali and Reddy 2000). In summary, the assay mixture was assembled in a 96-well flat-bottomed microtiter plate with each well containing 10% V8 juice, 3,000 animal spores and a series of double AgNP dilutions (100 to 0.10. µg ml −1, wt / vol) in 200 µl of total reaction volume. Control without AgNP. Each treatment was repeated four times, and experiments were repeated at least three times. Microtiter sheets are coated with parafilm and incubated at 25 ° C in a humid chamber to maintain high humidity. The optical density (OD 600 nm) of the microtiter plates was read immediately and 24 hours after starting the test with the Synergy H1 hybrid multi-mode microtiter plate reader (BioTek). Net growth was determined by subtracting OD 600 nm data at the beginning of the test from data at a 24-hour interval. Net growth data were standardized into untreated control and analyzed using a four-parameter sigmoidal logistic model using Prism 6.0 software (GraphPad Software, Inc.). Statistical analyzes of the suitability of the curves were also performed using Prism 6.0. The IC50 values, the AgNP concentration required to inhibit growth by 50%, were calculated from the equipped logistic curves. The plates are examined under a microscope and the image is recorded with a digital camera attached to an inverted microscope (IX8, Olympus).
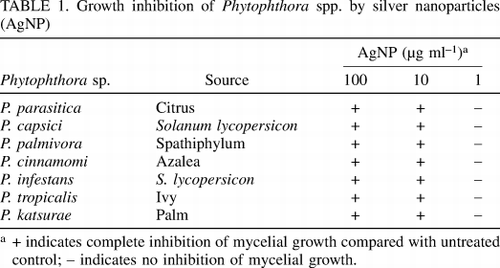
Table 1. Growth inhibition of Phytophthora spp. by silver nanoparticles (AgNP)
To evaluate the effect of silver nanoparticles on the germination of animal spores, the length of germ tubes and bags containing animal spores, animal spores were treated with two dilutions of AgNP (100 to 0.10 µg). ml -1, wt / vol) in the microtiter plates as described above. In addition, spore production and subsequent animal spore dispersion were regularly monitored for several days. Each treatment was repeated three times. Fifteen minutes after treatment with AgNP, swimming and saccharum animal spores were counted in three reverse microscopic field of view (IX8, Olympus). Ten hours later, three randomized photographs were taken in each well of the microtiter plate, and counted as germinated and non-germinated animal spores. The seed tube length was measured using the NIH Image J. software. Data on the germination dose response of animal spores, germ tube length and vesicles containing animal spores were fed into a four-parameter sigmoidal curve. numbers using Prism 6.0 software (GraphPad Software, Inc.). The IC50 values for the above parameters were calculated from the indicated fit curves.
2. Phytophthora inhibited in plants with silver nanoparticles
For the P. parasitica inhibition trials in planta, Nicotiana benthamiana (PI 555478) plants grown from greenhouse seeds were maintained at 25 ± 5 ° C with a cycle of 16 am / 8 pm. After 15 to 21 days, N. benthamiana plants were transferred to a walking growth chamber maintained at 25 ± 5 ° C with 16 h light (90 μmol / s / m 2) and an 8 hour evening cycle. Plants were sprayed until watery with the following treatments: AgNP at 100 µg ml −1, AgNP at 10 µg ml −1, mefenoxam (Subdue MAXX, 33.2 µg active ingredient ml −1), as a the positive control and water were the negative control. Each treatment consisted of three repetitions with each replication consisting of nine plants arranged in a 3 × 3 matrix in disposable plastic square pots (103 cm 2). One day later, the treated plants were thoroughly sprayed with 25 ml of P. parasitica animal spore suspension (10 5 animal spores ml -1). To prepare animal suspension, P. parasitica is cultured for 2 weeks on a 20% concentrated V8 20% juice agar containing 0.2% CaCO 3 under darkness at 25 ° C in a climate controlled growth chamber. . Spores are then scraped from the plate in 5 ml of sterile water and incubated at room temperature (25 ° C) for 30 minutes to release animal spores. In all, P. parasitica animal spores are treated with the same treatments in a microtiter disc for microscopic observation. After 10 days of calculation, numbers of healthy plants that survived and did not exhibit any Phytophthora disease symptoms were counted. Percent healthy plant data were computed for each treatment, and statistically analyzed for differences between treatments using the Student t-test. The experiment was repeated twice.
RESULTS
1. Synthesis and characterization of nano silver by UV-vis spectroscopy.
The change in color of the reaction mixture to golden brown or dark brown after mixing plant extracts and silver nitrate is a common feature of the biosynthesis of silver nanoparticles. Twenty-four hours after mixing A. absinthium aqueous extract with AgNO 3, a brown colloidal AgNP solution appeared (Fig. 1A). No change in color was observed with botanical extracts or with AgNO 3 alone under the same conditions. Visible UV spectroscopy also showed an increase in the UV-vis spectrum above 350 nm with the most pronounced increase in the 400 to 500 nm range (Figure 1B). AgNP is physically characterized using transmission electron microscopy, energy scatter X-rays, dynamic light scattering and zeta potential (Ali et al., Unpublished data).

Figure 1. Synthesis of silver nanoparticles using Artemisia absinthium extract. A, Glass vial marked with silver nitrate (AgNO 3), aqueous solution of A. absinthium and colloidal solution of nano silver (AgNPs), synthesized by mixing AgNO 3 and A. absinthium aqueous extract 1: 1. B, the absorption UV-vis (250 to 700 nm) of the above reaction mixtures after 24 hours of incubation.
2. Silver nanoparticles inhibiting Phytophthora spp. in vitro.
Ability to inhibit different Phytophthora spp species of nano silver. are evaluated in vitro at different stages of vegetative development and reproduction. These include growth of mycelium, germination of animal spores and germ canal length (vegetative growth), and the release of spores and mammals (reproduction), all of which are determine the ability to cause disease and develop disease. Effect of three concentrations of AgNP (100, 10 and 1 µg ml -1) on mycelium growth of different economically important Phytophthora spp. evaluated in vitro in microtiter plates. Microscopic examination of mycelium growth showed that AgNP applied at the rate of 100 and 10 µg ml -1 strongly inhibited growth of all Phytophthoraspp species. were tested after 12 hours of incubation (Table 1). Growth with AgNP 1 µg ml -1 treatment was similar to control.
To determine the efficacy and effectiveness of AgNPs, double series dilutions of AgNP ranging from 100 to 0.1 µg ml -1 were tested against P. parasitica and P. capsici. Dose response data were fitted with a logistic curve, indicating that AgNP inhibited growth of Phytophthora mycelia in a dose-dependent manner (Figures 2A and B). Conformity analysis showed that the R 2 is very high and the total standard error is low (Sy. ×, shown in Figures 2A and B), showing that the parametric match of the sigmoid curve with the data The dose response of AgNP was significant. The values IC50 and IC90, calculated from the dose response logistic curves, are very similar for P. parasitica and P. capsici, showed that AgNP inhibits both species equally well (Table 2). These observations were verified through microscopic examination of mycelium growth, in accordance with quantitative data (Figures 2C and D). Similarly, the minimum inhibitory concentration required for 100% inhibition of both Phytophthora species was 25 µg ml -1. Many antimicrobial compounds produce abnormal cell morphology such as irregularly shaped mycelium, over-branching, and fibrous mycelium. No such anomalous characteristics were observed in any Phytophthora spp. processed by AgNPs.

Figure 2. Inhibition of mycelium growth for Phytophthora parasitica and P. capsici with nano silver (AgNPs). The dose response curve displayed the growth inhibition of A, P. parasitica and B, P. capsici in response to different concentrations of nano silver. Light microscopy showing growth of mycelium of C, P. parasitica and D, P. capsici after 24 hours of treatment with the indicated AgNP concentration. Complete inhibition of both species was observed in response to 25 µg ml -1 AgNP. Thanh = 100 µm.
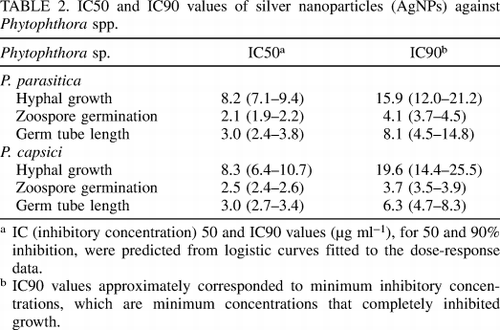
TABLE 2. IC50 and IC90 values of nano silver (AgNPs) against Phytophthora spp.
AgNPs decrease germination of animal spores, germ tube length and spore production in P. parasitica and P. capsici.
Successful inoculation of Phytophthora to plants depends on the germination of the animal spores and elongation of the seed canal. Therefore, the effects of nano-silver on these two pathogenic parameters were studied in vitro. Spore germination and seed tube length were both significantly inhibited by nano-silver in a dose-dependent manner. Similar to mycelium growth, the germination of animal spores and the seed tube length dose response data were also consistent with typical expression curves with highly consistent values (Fig. 3A). and B). IC50 and IC90 values for germination of animal spores and germ tube length were comparable for both Phytophthora spp. (Table 2). Similar to Imax (the maximum degree of inhibition achieved) for mycelium growth, germination of animal spores and germ tube length reaching 100% at doses of 10 to 25 µg ml -1 showed that AgNP synthesized in the current study is highly effective against Phytophthora spp. (Table 2).

Figure 3. Effects of nano silver (AgNPs) on different growth stages of Phytophthora spp. The dose-response curve shows the inhibitory effects of different concentrations of AgNP on germination of animal spores and the seed tube lengths of A, P. parasitica and B, P. capsici, and C, animal spore sac of P. parasitica.
The development of the Phytophthora epidemic depends on spore production and the release of animal spores. In in vitro assays, no spore production was observed for up to 15 days after silver nanoprocessing at the allowable mycelium growth rate (≥6.25 µg ml -1). At lower silver nanoparticles (≤3.12 µg ml −1), no mycelium growth was inhibited, spore production and animal spore production were not significantly different from the controls. process (data not displayed). Overall, these results indicate that AgNP synthesized with A. absinthium extract has strong activity against P. parasitica and P. capsici.
Treatment with AgNP enhances the spore tissue in P. capsici and P. parasitica.
Once released from the spore sac, Phytophthora animal spores swim around in water, usually for several hours, to find suitable sites of infection. After doing so, animal spores encapsulate, germinate, and enter the host tissues. During the normal microscopic examination of the cytoplasmic spores immediately following the establishment of the AgNP treatment experiments, we found fewer swimming animal spores after treatment with high concentrations of silver nanoparticles. than. This observation prompted us to hypothesize that nano-silver may be accelerating the mammalian encapsulation process. To test this hypothesis, we analyzed animal spore sacs at some point in time after treatment with different AgNP concentrations. Compared to the control, all mammals stopped swimming within 10 minutes of treatment with AgNP at concentrations ≥10 µg ml −1. AgNP-treated animal spores have a characteristic circular shape and quickly sink to the bottom of the microbial plates. In contrast, the swimming mammals observed in controls were not treated for at least 4 hours after starting the trial. These results were further confirmed by statistical analysis of swimming and marsupial data after 15 minutes of treatment with serial AgNP dilutions (100 to 0.10 µg ml -1). . Treatment with AgNP ≥1.56 µg ml -1 showed that the number of cystic animal spores was significantly higher than the control (Table 3). Significantly comparable results on animal spore sacs were reported for P. parasitica and P. capsici. Data on the dose response of marsupial and swimming animals were attached to sigmoidal curves (Figure 3C). These analyzes showed that the enhancement of spore formation in animal was dose-dependent with IC50 of 1.22 (95% confidence: 1.12 to 1.32) and IC90 of 2.79 (confidence interval). 95% confidence: 2.4 to 3.2). It is expected that the percentage of swimming animal spores decreases significantly as the AgNP concentration increases.
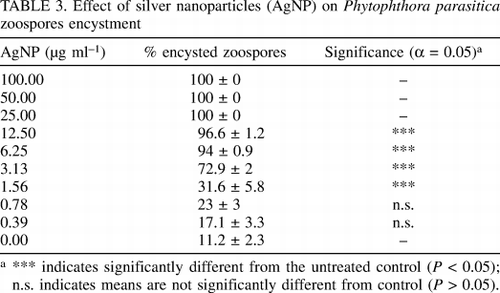
TABLE 3. Effects of silver nanoparticles (AgNP) on Phytophthora parasitica microspores
AgNP inhibits P. parasitica in planta.
To investigate the potential of nano-silver in controlling disease caused by P. parasitica in planta, tobacco plants were sprayed with 100 or 10 µg ml -1 AgNP, followed by inoculation of P. parasitica. Application with mefenoxam (SubdueMaxx, Syngenta), a fungicide commonly used against Phytophthora, and water used as positive and negative control measures, respectively. Similar treatments were performed in a micro dish to compare disease control in plants with in vitro growth inhibition. Microtiter disk microscopic data and visual observations of plants 5 days after P. parasitica infection are shown in Figure 4A. The percentage of plants that survived, without showing any symptoms of a Phytophthora fungal infection, was statistically analyzed. These analyzes showed that compared with a negative control, which displayed 7.7% of the mean plant survival, the AgNP treatments applied at 100 and 10 µg ml -1 showed the survival rate of the plant. cigarettes are 96.3% and 77.8% respectively (Figure 4B). In crop disease control with 100 µg ml -1 AgNP was comparable to SubdueMaxx (P = 0.42), while AgNP at a dose of 10 µg ml -1 was approximately 23% less effective than SubdueMaxx (P = 0.008), but fundamentally still (70%) better than the untreated control.

Figure 4. In plants, inhibition of Phytophthora parasitic on tobacco plants by nano silver (AgNPs). A, Tobacco plants are treated with indicated concentrations of AgNP and mefenoxam (SubdueMaxx) followed by culture of P. parasitica bacteria. The corresponding light microscopic image of the AgNP-treated mycelium in the test tube is shown in the row above. Pictures were taken 5 days after implantation. B, Bar graph showing percentage survival of plants after transplanting P. parasitica in response to treatment with AgNP, mefenoxam (SubdueMaxx) and water (control). Compared with water control, the survival rate of plants increased significantly in the AgNP treatment (*, P = 0.008, n = 27; **, P = 0.001, n = 27).
DISCUSSION
P. absinthium is an important medicinal plant with strong antioxidant activity (Ali and Abbasi 2014a, b; Ali et al. 2013). In this study, we have shown that nano silver synthesized by the water extract of this plant effectively inhibits some of the important agricultural Phytophthora spp. Many species in this genus cause destructive diseases in plants and they are known for developing fungicidal resistance and breaking down resistant genes by rapidly undergoing genetic mutations. The availability of broad spectrum protective silver nanoparticles will provide alternative tools for controlling diseases caused by Phytophthora spp.
Using chemical pesticides against plant microbiological diseases poses many challenges such as environmental pollution and the development of insecticide resistance in microorganisms. Therefore, the investigation to discover chemical pesticide alternatives against diseases caused by microorganisms is highly desirable. Nanoparticles that can be used as an alternative to chemical pesticides and studies reporting the use of nanoparticles to control fungi under field conditions are appearing in the literature (Kaur et al 2012; Panacek et al. 2009; Pimprikar et al. 2009). Most of these studies have focused on antimicrobial activity and to a lesser extent antifungal activities. To the best of our knowledge, no studies have been reported to explore the use of AgNP to control Phytophthora or any other species of oomycetes, in phylogenetic terms that are very different from real fungi. In this study, data showed a much lower concentration effect than 100 μg / ml for some species of Phytophthora spp. AgNPs are powerful and effective at various life stages, including mycelium growth, spore production and the germination of animal spores making them an excellent choice for testing. control Phytophthora disease. The in vitro MIC for AgNP synthesized in this study is approximately 25 µg ml -1. Using the same concentration, different inhibitions (24.7 to 83.5%) have been reported for AgNP synthesized against a number of different fungi (Kim et al. 2012), suggesting that AgNP reported in our studies may be more effective. However, this difference may be due to the source of AgNP (biological vs. chemical) or a difference in target organisms (Phytophthora vs. fungi).
Interestingly, treatment with AgNPs accelerated the formation of zoonotic pores in P. parasitica (Figure 3C). However, in contrast to the normal development of animal spores, including vesicle formation, germination and elongation of the germplasm, the growth and germination of vesicle spores caused by AgNP was Completely detained, suggesting that AgNPs may affect the normal physiological development of mammals. Further investigations are needed using genome, molecular and metaphysical studies to gain insight into how AgNPs affect the development of spores.
The majority of antibiotic studies using nanoparticles have focused on in vitro studies in a very isolated controlled environment that does not represent real world scenarios (Chernousova and Epple 2013; Prabhu and Poulose 2012; Prasad et al. 2011; Rai et al. 2009; Rai et al. 2012). To be useful for practical field applications, is a highly complex system that includes many physical factors such as temperature, humidity and light, and biological entities such as the community of microorganisms attached. For plants, it is imperative to evaluate the biological activity of AgNPs in plants. AgNP or any other nanoparticles that are active at the nanoscale, and their activity can be affected by various reducing and oxidizing agents in the micro-medium at the plant pathogen interface. In this study, AgNPs were very effective in disease control in tobacco plants cultivated with P. parasites under greenhouse conditions. The phylogenetic efficacy of AgNPs (100 µg ml -1) against Phytophthora reported in this study is very similar to that of AgNP for true fungal diseases such as pepper anthracnose and powdery mildew. on cucumbers and pumpkins in the field (Lamsal et al. 2011a, b). This match for efficacy against real fungi and Phytophthora spp., Placed in separate kingdoms (Judelson 2007), shows that nano-silver has broad-spectrum antimicrobial activity and that they can simultaneously control many diseases.
We did not observe any adverse effects on tobacco growth and anatomy even at concentrations many times higher than the minimum required for complete growth inhibition, suggesting that that the AgNP synthesized in this study is safe for application in plants. These results are consistent with several studies that have not reported any adverse effects of AgNPs when used at concentrations to control plant diseases (Lamsal et al. 2011a, b). However, our results differ from other reports, in that the phytotoxicity of AgNP is chemically synthesized (Gubbins et al. 2011; Kumari et al. 2009; Navarro et al. 2008). ; Stampoulis et al. 2009; Yin et al 2012). The discrepancy and similarity in results between our study and these reports may be due to different physical properties such as the limit of AgNP formed using plant extracts or other differences. differences in the methods of plant toxicity assay (Stampoulis et al. 2009). Although AgNP is used in a variety of consumer products, some reports indicate adverse effects of AgNP on mammalian cells (Ma et al. 2011), fish (Asharani et al. 2008), and crustaceans (Blinova et al. 2013). Based on the inconclusive reports above, future studies aimed at systematically investigating the ecotoxicity of the various AgNPs should be conducted before they can be commercialized for protect plants against disease.
CONCLUSION
The application of nanoparticles against plant pathogens is a growing field. In this study, we showed that the A. absinthium-mediated AgNP showed high potency against the important agricultural pathogens in the genus Phytophthora. In vitro treatments lead to complete suppression of P. parasitica and P. capsici at several stages of development including spore germination, elongation of the seed canal and spore formation. More importantly, in the application of AgNPs on tobacco plants, prevention of diseases caused by the fungus Phytophthora without any plant toxicity. The biosynthesis and use of AgNPs against Phytophthora can reduce the use of expensive chemical pesticides.
Reference source: Inhibition of Phytophthora parasitica and P. capsici by Silver Nanoparticles Synthesized Using Aqueous Extract of Artemisia absinthium
NANO NNA VIET NAM